 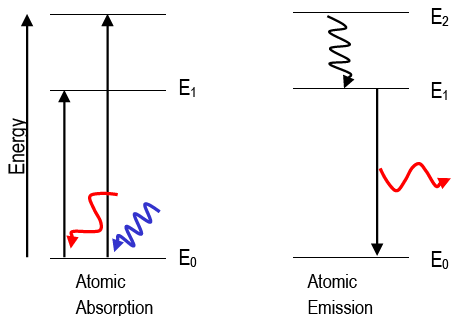
When a hydrogen atom absorbs a photon then the energy of the photon causes the electron to undergo a transition to a higher energy level (for instance n = 2 n = 3).The hydrogen emission spectrum generally comprises of radiations of distinct frequencies. When an electric discharge is passed through a gaseous hydrogen molecule, it was found that the hydrogen atoms present in the molecule dissociate thus leading to the emission of electromagnetic radiation by the excited hydrogen atoms. The hydrogen spectrum is a significant piece of evidence that further shows that the electronic structure of the atom is quantized. 
The emission spectra of molecules are commonly used in the chemical analysis of elements. This phenomenon also accounts for the emission spectrum termed as a hydrogen emission spectrum. Electrons present in an atom or a molecule absorb energy and then moves from a lower energy level to a higher energy level, on the other hand, electrons emit radiations when they come back to their original states.So the photon energy is equal to the difference in energy levels. If an electron is at an excited level ( E 1) and makes a transition to a lower level ( E 2), then the energy of the photon given out can be worked out with the equation: THESE ALLOW US TO IDENTIFY THE ATOMS IN A GAS. SINCE EACH ATOM HAS A UNIQUE SET OF ENERGY LEVELS, THEIR ABSORPTION AND EMISSION SPECTRA ARE UNIQUE. The electron has to get rid of the extra energy to do this and so it gives off a photon of light with a wavelength that corresponds to the energy level difference in the transition.įor a graphical depiction of this see the video below:Īnd emission spectra are simply the reverse! The electrons will drop back down (this may just be one energy level of multiple energy levels). This will leave black lines on the continuous spectra that correspond to the wavelengths absorbed.Īnd what happens to the electrons once they are excited? Well, only the photons that have a wavelength corresponding to the difference in energy levels will be absorbed (remember one electron can only absorb one photon). What will happen if you shine a spectrum of light of different wavelengths (continuous spectrum) on the atom? If this energy is equal to the difference in energy levels, the electron will be excited.Using either E = hf, E = hc/wavelength or for electrons E = 1/2mv^2 you can work out the energy of the incident photon/electron.Calculate the energy difference (in Joules) between energy levels.So the key to working out if this is possible is: If the energy is equal to the difference in 2 energy levels, the electron can be excited up 2 energy levels and so on. If a photon (or an electron) has energy equal to the difference in energy levels, an electron can absorb that energy to be excited to the next energy level. Exciting an electron – absorption spectra You will need to be able to convert between electron volts and Joules so make sure you get practice at this. Since we have 1V accelerating a charge of 1.6×10^-19 C then an electron volt is: 1eV = 1.6 x 10 ^-19 J You may have noticed that the energy of the energy levels is not given in Joules.Įlectronvolt definition: The energy an electron would have if accelerated through a potential difference of 1V. 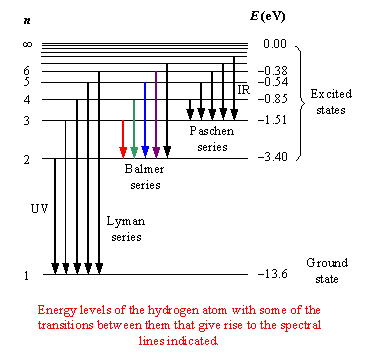
The calculations we do for both of these scenarios involving working out what the difference in energy is between the energy levels and comparing this to the energy of the incoming photon or high energy electron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
